This piece originally appeared on my Life Apps Brain & Behavior Blog on October 5, 2020.
It has been expanded on here.
Reward is a complex construct (for more see this excellent overview) but one reasonable definition is that reward refers to the fact that certain environmental stimuli have the ability to elicit approach responses, especially under biologically-based “need” conditions. Put another way: Stimuli that are desirable as a result of a biological need are “rewarding”.
Food is rewarding when we are hungry, water when we are thirsty.
Our brains are primed to learn about reward, specifically to learn what stimuli and actions in our environment will lead to obtaining outcomes that are necessary for survival - a process known as reinforcement learning. We learn certain behaviors are rewarding as they lead to us obtaining things we need to continue living.
| Reinforcement learning in its most basic form involves associating a stimulus with a response that then leads to a reward. This type of learning is done by virtually all animals. For example, a lab rat can learn that when a light comes on and it presses a particular lever in a specific environment it receives a food pellet. It learns light + press = reward via the following constituent parts: Light = stimulus Press specific lever after light = response Food pellet = reward And once this learned stimulus-response association is made, the stimulus itself can be perceived as “rewarding” in a process known as incentive salience (more on this later). |
Stimulus-response learning drives most of the behaviors we think of as “drug addiction” in people. When one is addicted to a drug of abuse, stimuli associated with the use of the drug (think, one’s neighborhood bar or a friend you routinely smoke with when together) can themselves drive drug use behavior. This is true even when the actual use of the drug is no longer “pleasurable” for the addicted individual. In fact, most drug addicted individuals do not find the use of their addicted drugs “pleasurable” any more.
This is because addiction is known to progress from a binge/intoxication stage of use to a withdrawal/negative affect stage and finally to a preoccupation/anticipation stage which can then reactivate drug use. Thus, drugs are initially used because they are pleasurable but over time this shifts and individuals use drugs of abuse to relieve negative withdrawal effects and not for pleasure. And, as mentioned above, drug use can be triggered by stimuli that were associated with drug use that promote preoccupation with using the drug even in individuals trying to stop or limit their use.
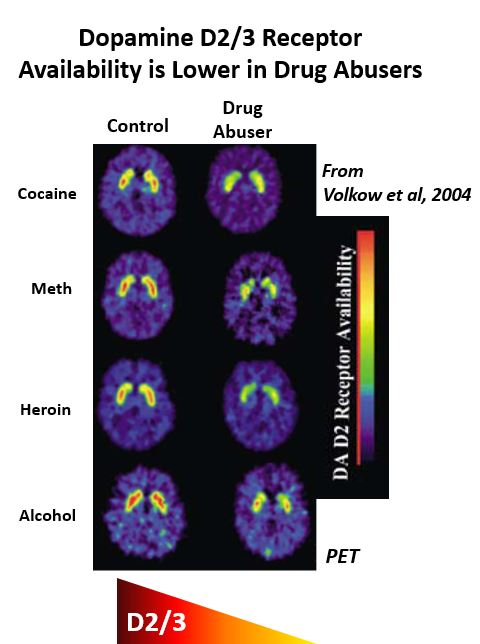
| Why does this happen? How can a drug that starts out as pleasurable lead to negative feelings of withdrawal when not used? Well, the brain is very adaptive and quickly modifies the biological environment such that there is less disturbance in dopamine (and other chemical) signaling after drug use. So, while drugs of abuse initially result in a large release of dopamine, this effect moderates with continued use. This change in responsivity to drugs of abuse is tolerance and explains why those addicted to drugs of abuse need to take larger and larger quantities to achieve the same effect. In fact, the continued use of addictive drugs results in notable changes in the brain dopamine system (see figure at right) which promotes a strong biological dependence on them. |
Much research has shown that dopamine does not signal “reward” (or to be more technical, pleasure) per se but rather is used in learning the various predictors of reinforcement in the environment - reinforcement learning.
This concept of dopamine signaling reinforcement learning was most famously demonstrated by the work of Wolfram Schultz, a professor at the University of Cambridge in the UK, who recorded the firing of dopamine-producing neurons (cells) in the brain of primates receiving reinforcing juice rewards.
Initially, these neurons fire to unexpected reward (juice) delivery. If a cue (tone or light) perfectly predicts the juice delivery (say 5 seconds before juice delivery), over repeated trials, Schultz found that the dopamine neurons fired in the presence of the cue (or, in psychological speech, conditioned stimulus) and not the reward. And when a reward is not followed by a stimulus previously paired with it, there is an observable dopamine “dip” locked to the time when the reward was expected to occur. See figure, below, from Schultz et al., 1997, illustrating reward prediction signaling in dopamine neurons.
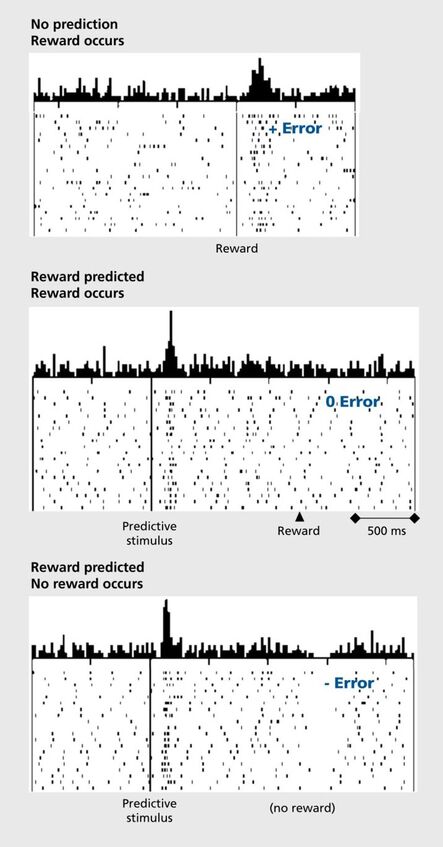
Dopamine signals predictors of rewards and not rewards themselves.
This concept of dopamine as a reward predictor has been extended to a hypothesis around the role of incentive salience in dopamine release and how this process can lead to craving or wanting behaviors in those addicted to drugs of abuse.
An amazing amount of work on this topic from Kent Berridge at the University of Michigan has demonstrated dopamine release is not associated with liking in the sense of a hedonic, pleasurable response but rather dopamine motivates behavior and affects how hard animals are willing to work for rewards (“wanting”).
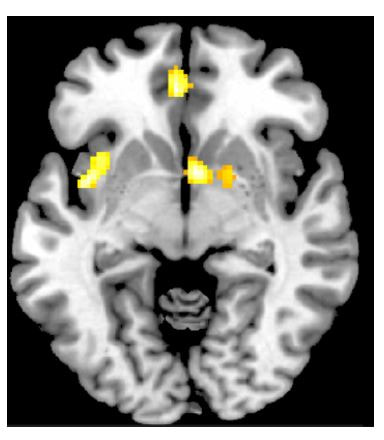
| My own work has sought to understand how the release of dopamine after oral d-amphetamine administration in healthy human subjects affects the brain. We found that dopamine release correlates with participants “wanting more” (NOT “liking”) d-amphetamine in three core brain regions often associated with reward and drug-related effects: ventral striatum (VS), ventromedial prefrontal cortex (vmPFC), and insula (see image at left). The VS is a core brain hub of reward valuation along with the vmPFC. Others have also found a relationship between VS dopamine release and “wanting”. The insula is a region of the brain often associated with drug craving/wanting and, in fact, damage to this part of the brain results in a loss of craving for cigarettes in smokers. Future efforts to modulate these craving-related systems and their associated dopamine signals through interventions such as transcranial magnetic stimulation may ultimately help drug-addicted individuals effectively stop their problematic drug use. |
Hopefully this piece has illustrated the complex role dopamine plays in signaling reward. Research that has emerged over the last few decades using sophisticated techniques to measure brain signaling in animals and humans has implicated dopamine in reinforcement learning processes and, by extensive the incentive salience of cues associated with rewards. The role of dopamine in signaling what stimuli predict reward is hijacked and pushed into overdrive by drugs of abuse that themselves release dopamine. Thus, after repeated pairings of stimuli and drug rewards, the brain adapts to respond powerfully to drug-related stimuli and cues, prompting craving in addicted individuals.
Not everyone is as susceptible to these dopamine-mediated learning processes, though. How individual differences in biology ultimately map onto risk for drug addiction is a matter of intense interest in the field of neuroscience but is beyond the scope of this current post. For the time being, I encourage you to explore the references below for more on the complex and nuanced role dopamine plays in reward and learning processes.
- A Neural Substrate of Prediction and Reward
- Neurobiology of addiction: A neurocircuitry analysis
- Liking, Wanting and the Incentive-Sensitization Theory of Addiction
- Pleasure Systems in the Brain
- Learning, Reward, and Decision Making
- Neural mechanisms underlying the vulnerability to develop compulsive drug-seeking habits and addiction
- Dopaminergic Mechanisms in Actions and Habits
- Imaging genetics and the neurobiological basis of individual differences in vulnerability to addiction




 RSS Feed
RSS Feed